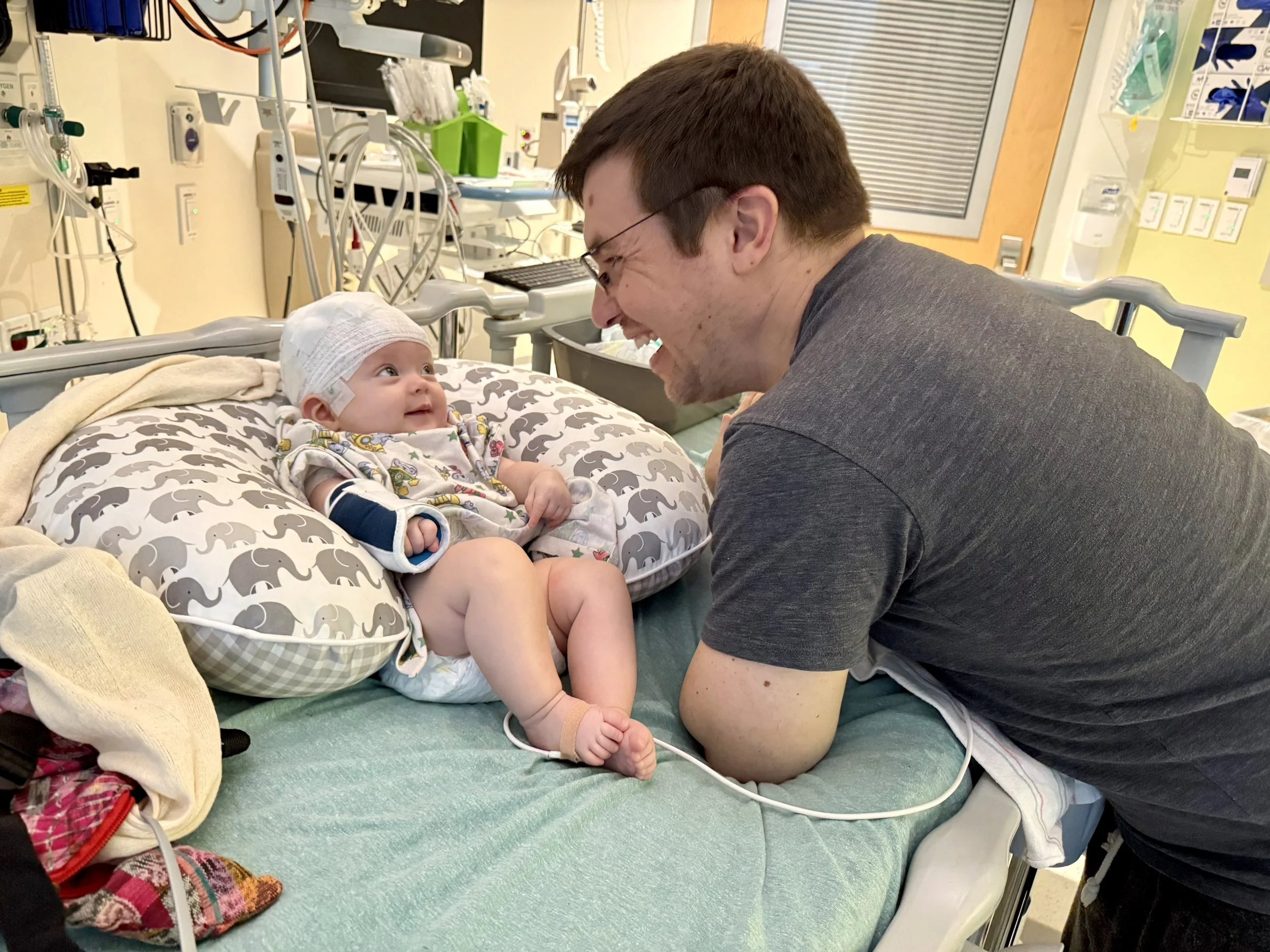
Ivy had her first seizure on March 13th, 2025, when she was three months old. She fell asleep in my arms—as she always did—and 10 minutes later, her eyes opened, her gaze fixed upwards, and her arms and legs stiffened and shook for what felt like the longest minute of my life. Within two weeks of that first seizure, she had two hospital admissions, an MRI, three EEGs, and a dozen tests to rule out infectious and metabolic problems. She also had a cheek swab for whole genome sequencing as part of a Boston Children’s Hospital study focused on rapid genetic diagnosis and tailored management of infant epilepsy.
While we waited for the results, Ivy stopped rolling over and vocalizing, her development stalled, and her seizures continued. On the eve of my first mother’s day, we got the genetic testing results that began to explain why. We learned Ivy has GRIA2-related neurodevelopmental disorder, caused by an ultra-rare mutation in a gene that helps build receptors for glutamate—one of the most important neurotransmitters for brain development, learning, and memory. From the 40 other people with GRIA2 disorder—and the one other child with Ivy’s exact mutation at the time she was diagnosed—we know that GRIA2 mutations cause cumulative and extensive harm: moderate to profound intellectual disability, limited or absent speech, major developmental regressions, and varying combinations of drug-resistant epilepsy, neurobehavioral disorders, movement and vision problems, sleep disturbances, and feeding difficulties.
Our plan since then has been clear: control her seizures and support her development while finding a way to address the underlying genetic mutation causing all of her symptoms. Thanks to precision medicine informed by her genetic diagnosis, Ivy was eventually started on an anti-seizure medication rarely used in children her age that has offered the seizure control she desperately needed. But the ongoing harms of this toxic mutation to her brain health are evident on EEG, and in her cognitive, motor, speech/language, and social/emotional delays. In animal research on similar mutations without intervention, brain cell death is observed that persists into adulthood. We are grateful to the multidisciplinary GRI Clinic at Children’s Hospital Colorado and researchers and advocates affiliated with CureGRIN for helping us understand the full impact of Ivy’s GRIA2 mutation.
As we write, at the start of this campaign, Ivy is 18 months old. She thrives in the calm and quiet of home, can’t get enough of her favorite musical toys, and can light up the room with her smile. Yet, many neurotypical children can say a dozen words by now, and Ivy hasn’t started babbling. She doesn’t clap or wave hello. Others are walking and learning to run, while Ivy crawls clumsily. Weekly physical, occupational, and speech therapies since she was 5 months old have definitely helped her make developmental gains, but these gains are hard-won and fragile and the neurodevelopmental harms of her GRIA2 mutation are ongoing and cumulative. Healthy glutamate functioning is the primary driver of neuroplasticity—the brain’s ability to adapt, grow, and form new neural connections throughout life. You can only grow so much on fragile roots, and the brain’s ability to re-root and re-grow is at its peak during early childhood. We have a narrow "developmental window" to intervene.
A personalized antisense oligonucleotide (ASO) treatment is Ivy’s only hope before this window closes forever. Like all of us, Ivy has two copies of the GRIA2 gene. One of her copies is healthy and working fine. The other copy has the toxic mutation. Because of how essential GRIA2 is for brain function, Ivy needs an “allele-specific” ASO that will just turn down the toxic GRIA2 copy and let the healthy copy do its job. This ASO is not a one-time repair of the typo in Ivy’s genetic code. It is a treatment that she will get injected into her spine every few months for the rest of her life to keep the toxic GRIA2 copy in check. Gene replacement and gene editing treatments may eventually be able to cure GRIA2 mutations like Ivy’s, but the scientific breakthroughs needed to bring those treatments to actual patients will take time. And time is the one thing Ivy doesn’t have.
Here’s where n-Lorem comes in. n-Lorem is a one-of-a-kind non-profit foundation dedicated to creating personalized ASO treatments for nano-rare patients—people with mutations that affect fewer than 30 people worldwide. n-Lorem was founded in 2020 and has already treated 50 patients with ASOs, with incredible results and a spotless safety record. One of n-Lorem’s youngest patients started receiving her ASO treatments when she was three years old. Among other big gains, she went from not speaking to speaking 2-3-word sentences within six months of starting treatment.
n-Lorem has already given us something we thought we might never find: a clear path forward. Not hope for treatment someday. But a proven pathway to a life-changing treatment before Ivy’s developmental window closes forever. Help us reach the finish line. Your donation is not just funding a treatment, it’s funding innovative precision genetic medicine for ultra-rare disease, scientific advances that will benefit the broader GRIA2 and GRI disorder community, and a future of flourishing for Ivy.
Emily, Ivy’s mom
Will, Ivy’s dad